Biologics Safety Testing Market: Global Industry Analysis, Trends, Market Size and Forecasts up to 2023
The report on
global biologics safety testing market provides qualitative and quantitative
analysis for the period of 2017 to 2023. According to report the global biologics safety testing market is expected
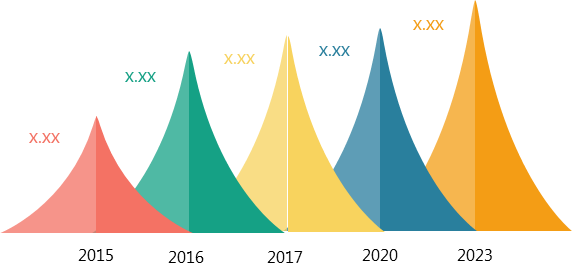
to grow at a CAGR between 12.0% and 12.5 % over the forecast period of 2017 -
2023. The global market size was USD 2.19 billion in 2015.
Figure
1: Global Biologics Safety Testing Market by Value 2015 - 2023
Market
Insights:
The report
identified that global biologics safety testing is driven by factors such as number
of drug launches are increased, technological advancements, increase in biotechnology and pharmaceutical
industries driven by government support and regulations. While the restraining factors include high cost of biologics safety testing. Further,
the report identifies the opportunities in the world market as emerging markets
offer profitable growth opportunities, increasing
pharmaceutical outsourcing.
Segments
Covered
The report on
global biologics safety testing market covers the segments based on instruments
and services, test type, and application. The instruments
and services
of biologics safety testing include instruments, kits & reagents and
services. The test type segment includes bioburden tests, endotoxin tests,
adventitious agent detection tests, cell line authentication and characterization
tests, residual host contaminant detection tests, adventitious agent detection
tests and other test. On the basis of application the
global biologics safety testing market is segmented as vaccine & therapeutics
development, tissue
and tissue-related products testing, blood and blood-related products testing, stem
cell research and cellular and gene therapy.
Geographies
covered
The report provides
regional analysis covering geographies such as North America, Europe,
Asia-Pacific, and Rest of the World. In this section the key trends and market
size for each geography are provided over the period of 2017 – 2023.
Figure 2: Global Biologics Safety Testing Market by region
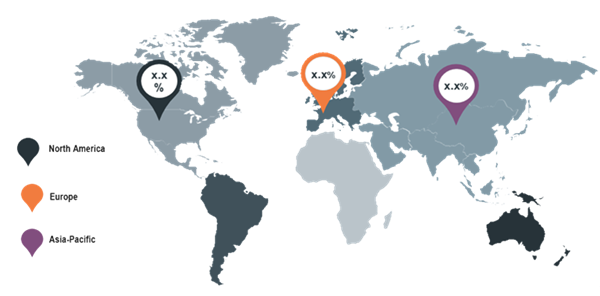
Globally, North
America
market dominated the world biologics safety testing market followed by Europe
and Asia-Pacific. Furthermore, Asia Pacific
market is
expected to grow at the highest CAGR over the forecast period 2017-2023.
Due
to the increasing emerging economies like china and India, growing
population.
Companies
Profiled:
The
report provides profiles of the companies in the global biologics safety testing
market such as, Toxikon Corporation, Charles River Laboratories International,
Inc., Avance Biosciences Inc., Thermo Fisher Scientific Inc., Merck KGaA, Lonza
Group Ltd., Pace Analytical Services Inc., Eurofins Scientific Se, Wuxi Apptec,
Sartorius AG, Cytovance Biologics, Inc.
Report
Highlights:
The report
provides deep insights on demand forecasts, market trends and micro and macro
indicators. In addition, this report provides insights on the factors that are
driving and restraining the global biologics safety testing market. Moreover,
IGR-Growth Matrix analysis given in the report brings an insight on the
investment areas that existing or new market players can consider. The report provides insights into the market
using analytical tools such as Porter’s five forces analysis and DRO analysis
of biologics safety testing market. Moreover, the study highlights current
market trends and provides forecast from 2016 to 2023. We also have highlighted
future trends in the biologics safety testing market that will impact the
demand during the forecast period. Moreover, the competitive analysis given in
each regional market brings an insight on the market share of the leading
players. Additionally, the analysis highlights rise and fall in the market
shares of the key players in the market.
